Description
For monitoring performance parameters of anitbiotic residue test kits.
Storage
You’ll need to keep your Delvotest® Milk Controls in a cool, clean and stable environment (2–8°C, away from light). We advise leaving them unopened in the original packaging until ready for use.
Preperation
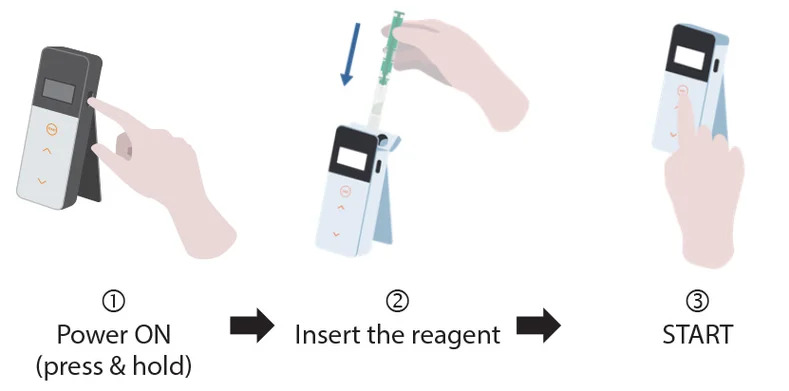
To ensure accurate results, it is essential to prepare an entire vial at once — we do not advise partial preparation. As Delvotest® Milk Controls are lyophilised, dry comingled milk, we usually refer to the process of preparing them as ‘reconstituting’. To get your controls ready for use, just follow these steps:
- Wash your hands thoroughly and clean your workspace. Avoid using strong detergents or disinfectants, as they could interfere with their performance.
- Tear off the aluminium seal.
- Carefully remove the ‘stopper’ from the bottle and place it upside down somewhere to avoid contamination.
- Using a clean pipette, add 15 ml (± 0.5 ml) of distilled or demineralised water. Water used for preparing standard reagents must meet ISO 3696 Grade 3 or higher specifications. This ensures low levels of ionic and organic contaminants and supports consistent analytical performance. Avoid using tap water or water of unknown purity.
- Put the ‘stopper’ back on.
- Hold the ‘stopper’ in place and gently invert the vial c. twenty times to mix the contents thoroughly — avoiding creating foam, and ensuring the solution is homogeneous. Make sure that an homogeneous solution is obtained. Otherwise proceed with mixing till all particles are dispersed. If needed, use a mechanical stirrer.
- Allow the contents to equilibrate at room temperature for c. 30 minutes.
- Remix gently before use, inverting the bottle c. twenty times.
Once reconstituted, your Delvotest® Milk Controls should be used only on the same day of reconstitution.
Freezing for Future Use
Reconstituted Delvotest® Milk Controls can be portioned into smaller volumes and frozen for later use. Be sure to portion the controls into the smallest practical volumes depending on your use case for future use to avoid unnecessary waste. To prepare and store them properly, just follow these steps:
- Directly, without delay, after reconstituting as descripted above.
- Gather clean vials of an appropriate size for portioning.
- Label each vial with at least the following information:
- Control type including species (e.g., ‘Cow Positive Pen G 4ppb’).
- Batch number.
- Today’s date.
- Two months from the current date, as reconstituted controls can be stored for up to two months in the freezer.
- Operator’s name / ID
Altogether, your vial’s label notes should look something like this:
Cow Positive Pen G 4ppb
Batch: 23D19/6
Prepared on: 7 July 2024
Valid until: 7 September 2024
Prepared by: Jan Kees
- After reconstituting the control, pipette the required amount (e.g., 2 ml) into the vials, close them tightly, and place them directly into a freezer at c. -18°C.
- Follow the ‘Preparing for Use’ steps in the section above — keeping in mind that on the day of re-use, you’ll need first to thaw the vial by placing it in lukewarm water for a short time (c. 5 mins), mixing the contents thoroughly before using.
Once thawed, the reconstituted controls can only be used on the same day if stored between 2–8°C (or on ice). Do not refreeze the controls after thawing.
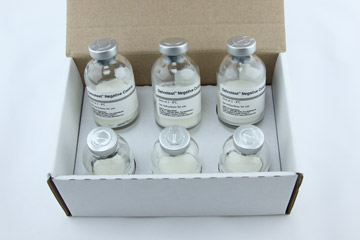
Pack Size
6 vials
If Negative Control Tests Positive
A negative control should never test positive. If it does, there are several potential reasons to investigate:
- Contamination. Contamination during sample preparation or testing is the most common cause of a false positive. Ensure proper sterilisation and handling techniques.
- Storage Issues. Improper storage of the control can lead to degradation. Verify the control was stored in 2–8°C, away from light.
- Reagent Quality. Ensure that all reagents used in the test have not expired and have been stored properly.
- Instrumentation Issues. Perform regular maintenance checks on your equipment and validate the test performance periodically to avoid false positives.
Once you’ve identified the likely cause, take corrective action by repeating the test with fresh negative controls, validating your equipment, and improving handling practices.















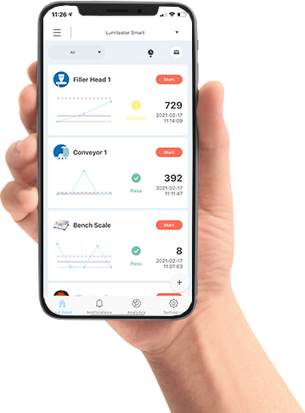
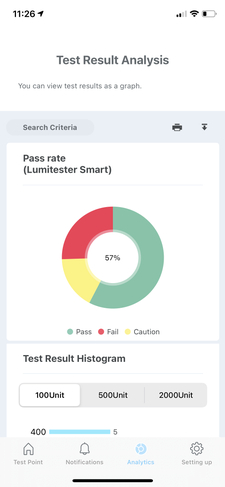 Exportable Data and Reports for Regulatory Compliance
Exportable Data and Reports for Regulatory Compliance
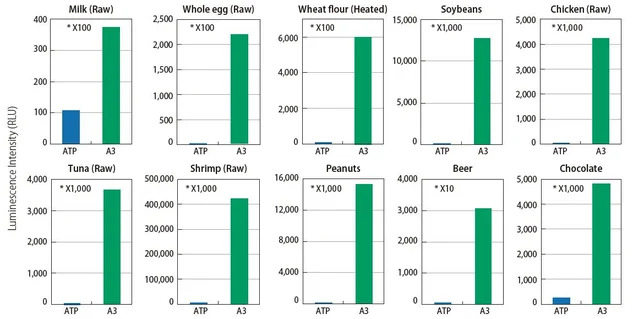 See how A3 technology finds what ATP technology can’t
See how A3 technology finds what ATP technology can’t